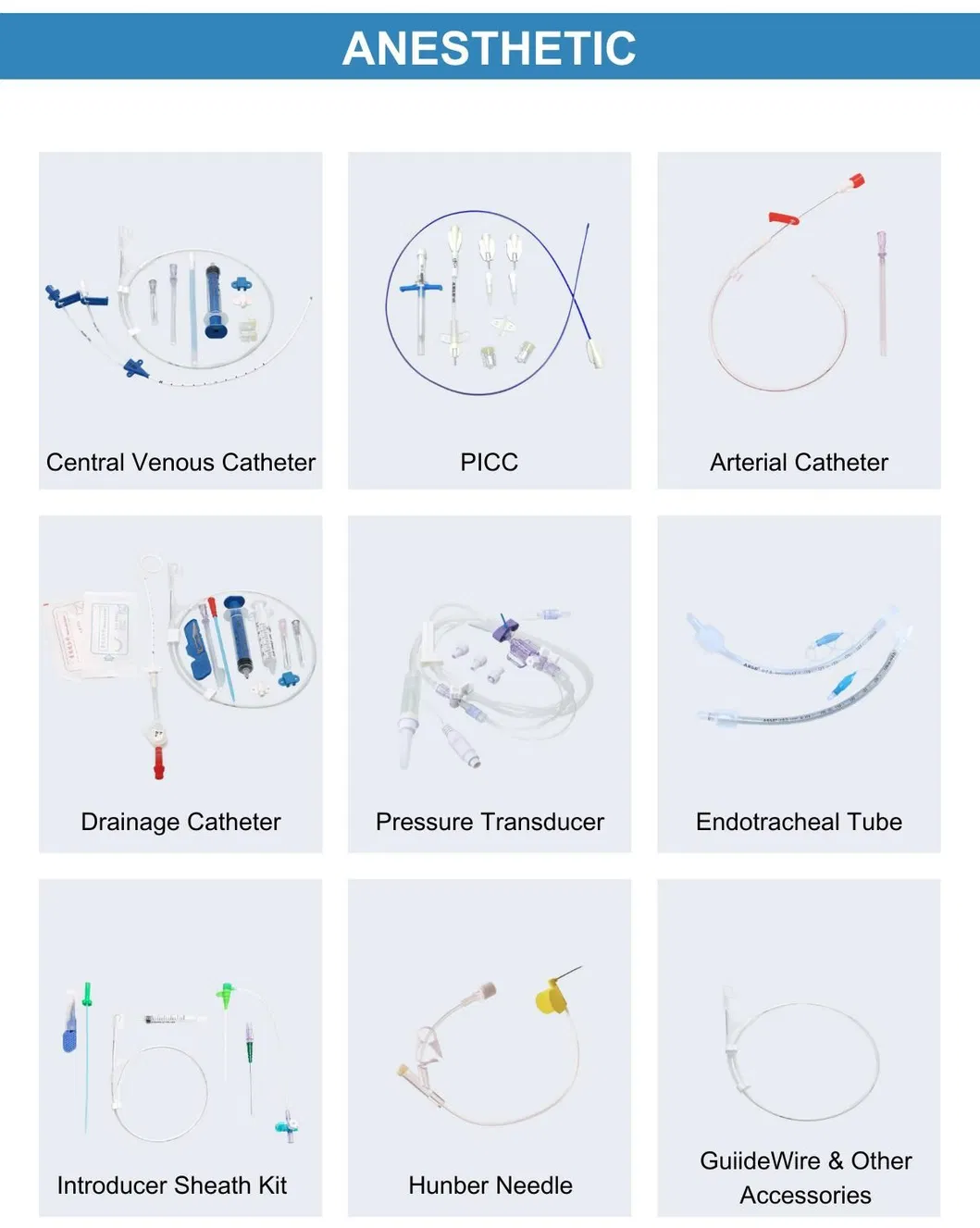
Overview & Basic Info
Sterilization
Ethylene Oxide (EOS)
Safety Standard
ISO 10993
Size
Triple Lumen 7fr*20cm
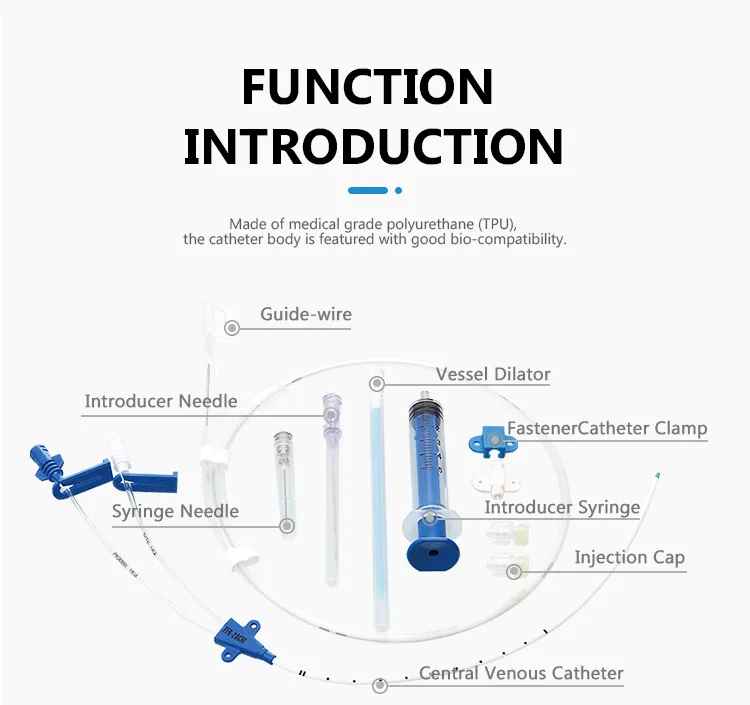
Product Description
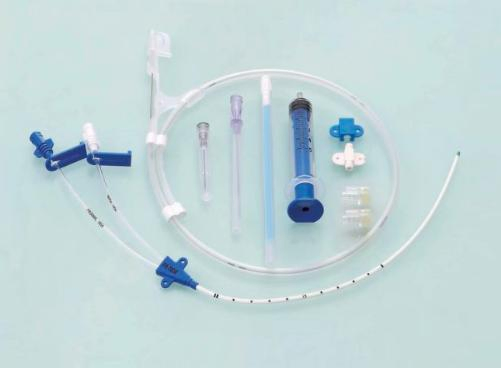
Central Venous Catheter CVC Kit
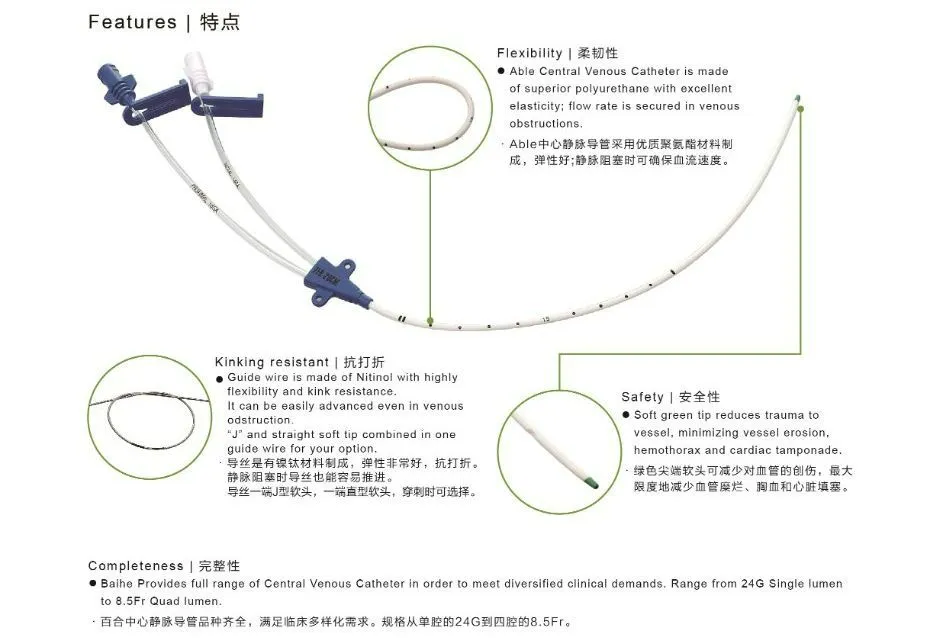
Features & Benefits:
- Movable Clamp: Enables secure anchorage at the puncture site, minimizing trauma and irritation regardless of catheter depth.
- Precise Placement: Depth markings ensure accurate positioning from either the right or left subclavian or jugular vein.
- Soft Tip Design: Significantly reduces vessel trauma, minimizing risk of erosion, hemothorax, and cardiac tamponade.
- Superior Radiopacity: Facilitates easy confirmation of catheter and multi-lumen tip placement under fluoroscopy.
- Vessel Dilator: Ensures "super soft" catheters are easily placed percutaneously, enhancing procedural efficiency.
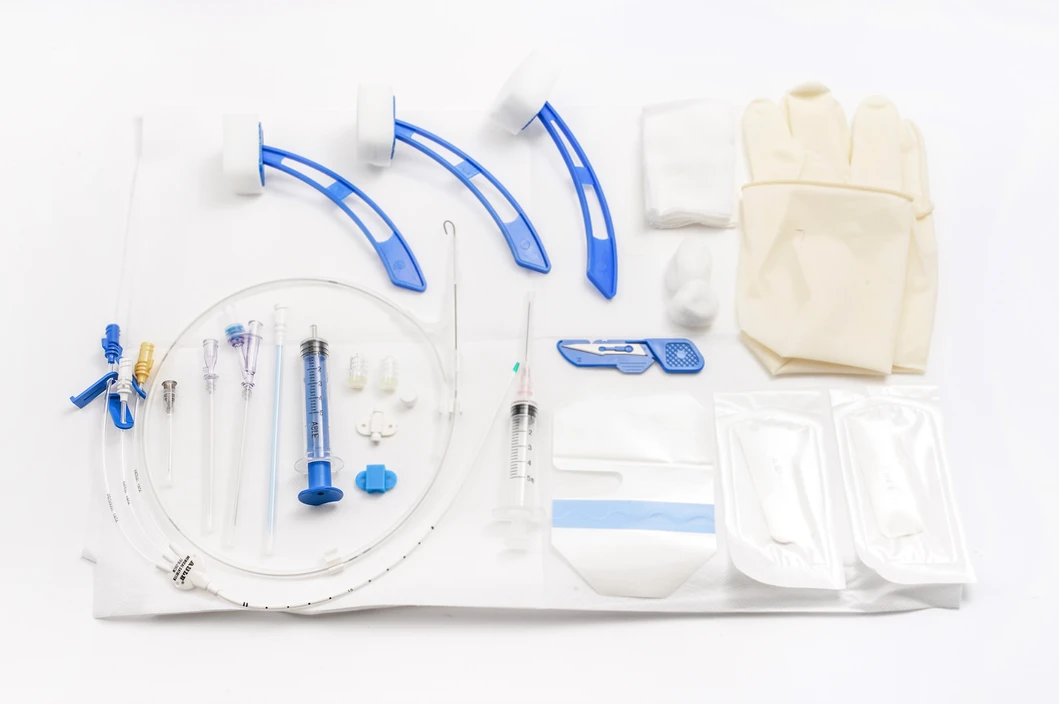
Detailed Product Display
Technical Configuration Remarks:
Adult CVC kits are available in both straight and Y-shaped introducer needle configurations. For Y-shaped options, the letter 'Y' is added to the product code (e.g., 'FC32726Y').
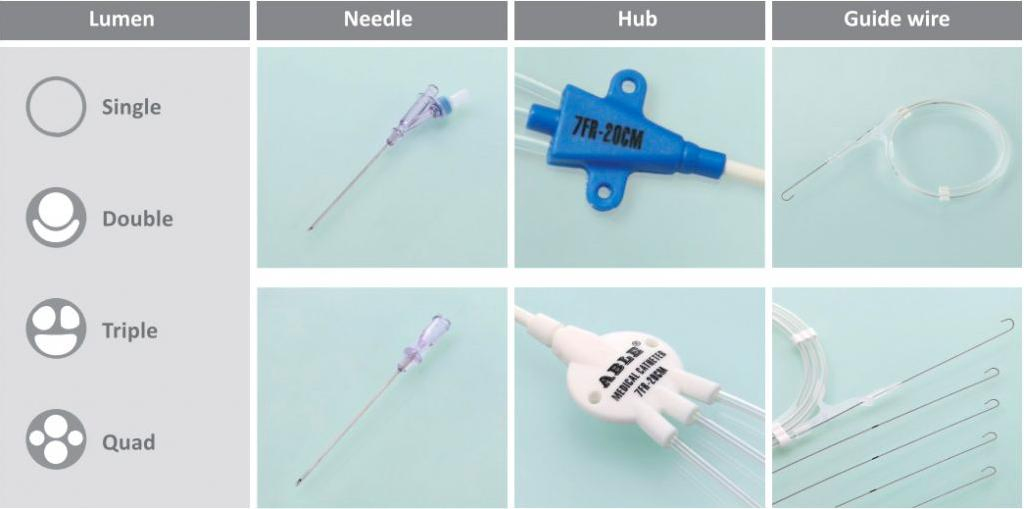
Available Ranges:
- Pediatric: Single lumen (24Ga-18Ga), Double lumen (4Fr, 5Fr), Triple lumen (4.5Fr, 5.5Fr).
- Adult: Single lumen (16Ga, 14Ga), Double lumen (7Fr-8.5Fr), Triple lumen (7Fr, 8.5Fr), Quad lumen (8.5Fr).
- Hub Designs: 'FC' indicates triangular hub; 'FV' indicates cylinder or round hub designs.
Professional Production & Quality Control
Focus on quality, respect on life! Our production environment adheres to strict medical standards to ensure patient safety and product reliability.
Our Advantages
Packaging & Transportation

Frequently Asked Questions
Q1: What is the primary sterilization method for the CVC kits?
A: All Central Venous Catheter kits are sterilized using Ethylene Oxide (EOS) to ensure medical-grade safety.
Q2: How does the soft tip design benefit clinical procedures?
A: The soft tip significantly reduces vessel trauma, lowering the risk of complications such as hemothorax or cardiac tamponade during placement.
Q3: Are there options for different hub designs?
A: Yes, we offer triangular (FC code), cylinder, and round hub (FV code) designs depending on the lumen configuration.
Q4: Can the placement of the catheter be easily verified?
A: Yes, the catheters feature superior radiopacity, allowing for clear confirmation of the tip's position under fluoroscopy.
Q5: What sizes are available for pediatric patients?
A: We offer a range for pediatrics including single lumen (24Ga to 18Ga), double lumen (4Fr, 5Fr), and triple lumen (4.5Fr, 5.5Fr).
Q6: What is the shelf life of the sterilized kits?
A: The products come with a 3-year quality guarantee and shelf life from the date of manufacture.


 Nordi Medical
Nordi Medical