




1 / 5
 Nordi Medical
Nordi Medical










| Still deciding? Get samples of $ ! US$ 0.05/Piece Request Sample |
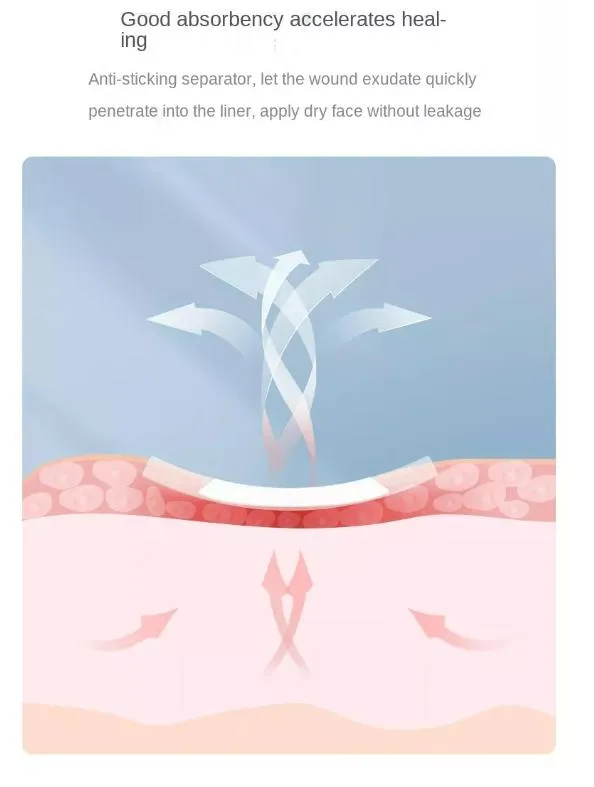
Sterile self-adhesive island dressing is crafted from nonwoven fabric or highly permeable polyurethane (PU) film, coated with medical-grade acrylic adhesive and an absorbent fabric pad. It creates an optimal occlusive environment for wound healing while remaining breathable.
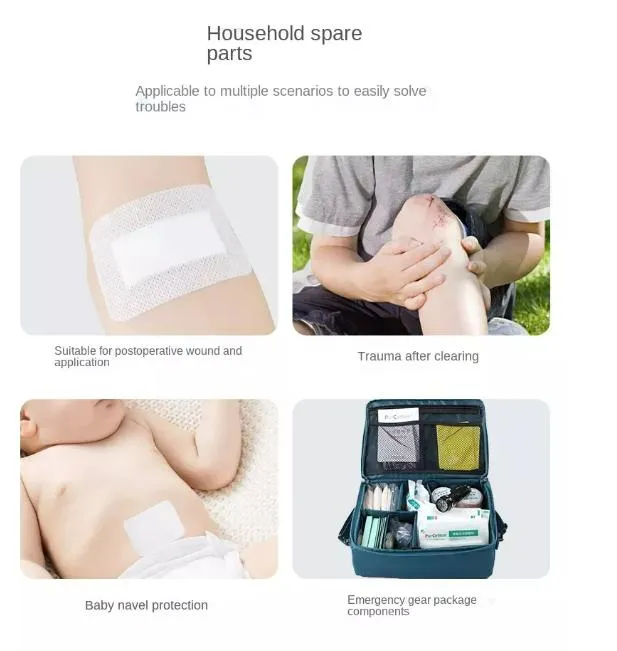




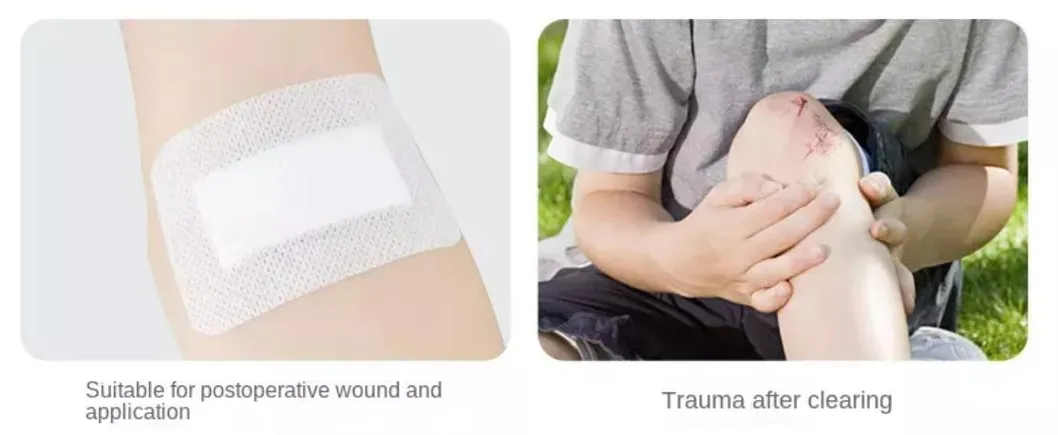
Designed for professional care across various wound types:

As a leading professional manufacturer of medical disposable products since 1989, we maintain the highest standards in the industry.


